Welcome to Our Blog
Last chance to register to the PG Clinical Research Certification and On-boarding Program CRP3.0 with the 2021 fees
Last chance to register with the 2021 fees to the new expanded PG Clinical Research Certification...
Invest in your future, vaccine clinical trials need 10 times more staff, the job market is red hot.
Why it is the best time now to join the rewarding clinical trials industry The clinical trials...
Black Friday gives a last chance to get an affordable access to well-paid careers in the booming clinical trials industry. PG Diploma with 3 remote internships and 7/7 placement support till hired.
Last chance to get affordable access to well-paid careers in the booming clinical trials industry....
Want to work in Canadian clinical trials? Network with the Clinical Research Association of Canada (CRAC) to ask them LIVE all your questions
Do you want to work in the Canadian clinical trials industry? Join our free networking meeting by...
Free networking meeting on Nov. 24 with Rene Ategwa, SrCRA and Clinical Team Lead at PPD who will answer LIVE all your questions
The international Clinical Research Academy, ICRA (The CRA School of Montreal) invites nursing and...
Job offer: Administrative Assistant, full time, remote, functionally bilingual
JOB OFFER Position: Clinical Research Administrative Assistant (functionally Bilingual), full-time...
APPROVAL OF MEDICAL DEVICES
THE MEDICAL DEVICES APPROVAL PROCESS Medical devices are non-metabolic and...
CRA-SCHOOL GLOBAL CLINICAL RESEARCH SERIES #1, UK Chapter
On Oct.29, Monir El Azzouzi, CEO of Easy Medical Device, UK, will discuss the role of the clinical...
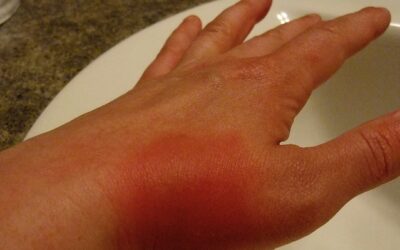
Adverse Drug Reactions
Adverse Drug Reactions Ranging from mild, unnoticeable effects to severe anaphylactic...
The Importance of Adapting Your CV for Each Application in clinical research
Many people have “their CV” and send it out for any and all positions that they are interested in....
The Medicago plant-based Covid-19 Canadian vaccine, Clinical Safety and Pharmacovigilance
The unique Medicago plant-based Covid-19 Canadian vaccine Clinical Safety &...
The Routine Monitoring Visit and ISF Review, Training Workshop with Ken Boudrault, SrCRA
The Routine Interim Monitoring Visit and ISF Review Detailed analysis of challenges in a pandemic...
To enroll with the CRA School Montreal, please fill in the application form by clicking on the ‘Enroll Today’ button.
Clinical Research Advisor
Ken Boudreault,
Senior CRA II
Synteract, Montreal
Consultant & Mentor
20+ years experience
Data Management Advisor
Nassima Benmouffok
Ass. Director of Data Management,
Ethica CRO, Montreal
Senior Data Manager
Medical Science Liaison Advisor
Thomas Rolain,
PhD, CCRP/CCRA
Syneos Health
SrCRA and MSL
Pharmacovigilance and Drug Safety Advisor
Medical Writing Advisor
Sefika Ozturk, PhD
Medical advisor
MSL at Merck
Clinical Trials Management Advisor
Radia Ouelaa, PhD
Clinical Team Manager, PPD
Onco/Hematology
Clinical Research Management
Janet Vergara, SrCRA
Ass.Director at Merck
Clinical Research Manager
Clinical Trials Advisor, Trainer and dedicated Mentor
Bahareh Najjar SrCRA
MedPace, Montreal
CRA Mentor and Trainer
Clinical Operations Advisor
Anna Leonov, PhD
Clinical Operations
Syneos Health
Regulatory Affairs Advisor and Mentor
Mona Ramezani
IH SrCRA at Merck
Regulatory Affairs
Medical Devices Advisor
Monir El Azzouzi,
CEO and Consultant
Easy Medical Device
Medical Writing Advisor and dedicated Mentor
Dr Anand Devasthanam, PhD
Ass. Medical Director
Syneos Health, Boston
France
Karima Kezadri, MBA, MSc, CCRP
Regional Rep. for
Maghreb & MENA
Tel: +213559918058
hopekarima@yahoo.fr
Nigeria
David Dele-Davids
Lagos, Nigeria.
Tel:+2349093309249
WhatsAp: +19495202173
deledavidsconsulting@gmail.com
India
Monisha Sundar
Ontario; Canada
New Delhi; India
Tel: +16472387293
+918427688701
mishu_shiv@hotmail.com
Bulgaria
Dr. Jamil Rashed
Sofia, Bulgaria.
Tel: +359879450847
Djamilrashed@gmail.com
AUSTRALIA
Jyothi Surada,
BPharm, CCRP
CRC,
BC
Benard Kimwei
Burnaby, BC
Tel: +1 403 465 6900
benard.kimwei@gmail.com
AB
Olaniyi Taiwo
Edmonton, AB
Tel: +1 306 450 2974
sotaiwo06@gmail.com
MB
Dina Tabatabaei, MA,
CCRP, CRA School Rep
Brandon, MB
(416)879-7575
dtabataba88@gmail.com
USA
David Dele-Davids
Baltimore, MD, USA.
Tel: +1(949)520-2173
deledavidsconsulting@gmail.com
UGANDA
Daniel Okodan, MSc,
CCRA, CRA at PPD
Entebbe, Uganda
okodandaniel@gmail.com
DUBAI
Taline Sagherian, RD
CCRP, Dubai, UAE
+(971) 50 935 4208
sagheriantaline@gmail.com
FIJI
Oluwafemi Ojo, MD, CCRA
Physician, Tawua, Fiji
drfemiojo@gmail.com
GERMANY
UGANDA
Sylvia Namanda,
BSN, CCRP,
Makarere University
(+256) 756 762 005
sylvian531@gmail.com
IVORY COAST
ISSATOU DIALLO, CRA
Regional Manager
Afrika at MCT-CRO
issatoubella@gmail.com
Medical Writer
Christina Sanguinetti,
Medical Writer, CEO
LaconicMed, ON
christina@laconicmed.ca

Workshops
405 Avenue Ogilvy, unit #101
Montreal, QC, Canada H3N 1M3
(legal address, no admission)
Contact Us
info@cra-school.com
Information : +1 (514) 534-0273
Support 7/7 : +1 (514) 257-3003
By Skype: CRA School Montreal
- © 2009 - 2024 CRA-School - International, All Rights Reserved.